Our research group focuses on the development and application of advanced light imaging methods, particularly quantitative phase microscopy (QPI). We belong to the field of biophotonics, which combines optics, physics, and biology and studies the interaction of light with living systems. We develop optical methods that enable non-invasive real-time observation of cells and tissues, and we also participate in teaching biophotonics to medical and technical students. QPI uses the light phase as a source of information and allows live cells to be observed without staining, chemical treatment, or stress on the sample. This provides highly sensitive data on the mass, structure, and dynamics of cells in their natural environment. We study not only biological samples, but also the interaction of light with complex structures. We focus on how light changes as it passes through cell tissue or nanostructures—for example, by measuring phase delays that reflect local changes in the refractive index or density of the material. We also analyze polarization changes and anisotropy, i.e., different optical behavior in different directions. These optical signals provide detailed information about the internal organization, morphology, and physical properties of the biological and nanotechnological systems under study. Our team consists of physicists, biologists, engineers, and students. We combine expertise in optics, instrumentation, and biomedicine to develop non-invasive imaging techniques and create tools that contribute to a deeper understanding of living systems - fast, precise and without harming the sample
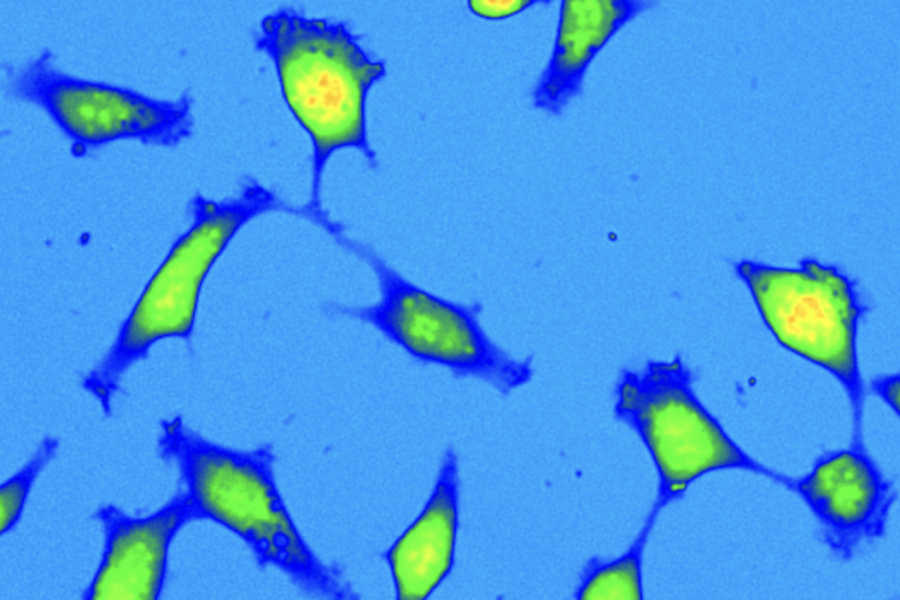
Quantitative phase microscopy allows live cells to be observed in real time without the use of dyes, eliminating the phototoxicity and distortion typical of fluorescence techniques. The method provides accurate information on biomass quantity, growth, division, and cell movement, and allows processes that are difficult to measure using other imaging techniques to be captured. QPI plays an important role in oncology. Fresh tumor cells from biopsies often respond differently to drugs than normal cell lines. Using QPI, it is possible to monitor in detail the migration rate, invasiveness, and response to cytostatics directly in living cells. Thanks to its high sensitivity, we can distinguish even very subtle differences in cell behavior, which may be crucial for future personalized therapy. Our technologies also contribute to materials research. They enable detailed analysis of the properties of nanostructures and metasurfaces that modify light in completely new ways. These findings can fundamentally influence the design of advanced optical elements and expand the possibilities of modern microscopy and photonics

We are behind the development of the Q-Phase holographic microscope, which was created based on our long-term experimental and theoretical knowledge in the field of optics and biophotonics. This device combines high resolution, low light load, and accurate light phase measurement. In collaboration with TELIGHT, Q-Phase is now manufactured and used in laboratories around the world. We have also developed a methodology for observing live tumor tissues from biopsies. It allows us to monitor cell behavior directly at the edge of the tumor — i.e., in areas where there is a risk of invasiveness. Our experiments have confirmed fundamental differences between fresh tumor cells and long-term cultured lines, which has a significant impact on the evaluation of anticancer drugs and their effects. We have long contributed to the development of optics and holography. Our research into incoherent light has enabled us to suppress artifacts typical of laser systems and increase measurement accuracy. By combining phase imaging with fluorescence, we have created multimodal approaches that provide a comprehensive picture of cell mass and specific structures. Our technologies have a real impact in biomedicine, drug development, and advanced materials research

We are currently working on a new generation of quantitative phase imaging, which will enable 3D reconstruction of biological samples with higher quality than current systems provide. We are developing software and optical approaches that will enable the reconstruction of sample volume from phase data and the imaging of tissues or organoids in greater depth and complexity. We are also investigating the effect of drugs on tumor cell migration. Some already approved drugs have secondary effects that may limit cell migration. Using Q-Phase, we evaluate their effect on living cells in detail, contributing to the identification of potential anti-metastatic therapies. In the field of materials science, we focus on nanostructures and metasurfaces that can control light propagation at the wavelength level. We collaborate with teams that design and manufacture these structures and use our microscopy to verify their functionality. We also use the knowledge gained to develop new optical solutions. The recent addition of a fluorescence module to the Q-Phase microscope allows us to combine non-invasive phase data with specific labeling of cell structures. This gives us a more comprehensive view of biological processes and significantly expands the possibilities of multimodal imaging. At the same time, we are developing methodologies for observing biopsies, including procedures for working with thicker tissue sections, which until now could not be phase-imaged
