5. Aug. 2021
Researchers reporting in The Journal of Molecular Diagnostics have developed the LYmphoid NeXt-Generation Sequencing (LYNX) panel that analyzes both standard and novel molecular markers in the most common lymphoid neoplasms
Philadelphia, July 29, 2021 – Increasing knowledge about genomic biomarkers has facilitated better monitoring and personalized management of patients with B-cell malignant tumors. A new integrative, capture-based, next-generation sequencing (NGS) panel, LYmphoid NeXt-Generation Sequencing (LYNX), can detect and analyze standard and novel biomarkers in the most common lymphoid neoplasms simultaneously. This represents a crucial step towards more effective personalized treatment of these diseases and facilitates further research, report researchers in The Journal of Molecular Diagnostics.
Genomic profiling by NGS provides novel critical clinical information about prognostic and predictive biomarkers. NGS studies have identified several genomic alterations in hematologic malignant tumors, which have improved our understanding of the disease course as well as the evolution of these neoplasms.
“With a constantly growing number of genetic markers with evidenced or potential clinical impact in lymphoid neoplasms, a more comprehensive genomic test is highly desirable,” explained lead investigator Prof. Dr. Sarka Pospisilova, from the Faculty of Medicine, Masaryk University and University Hospital Brno; and Central European Institute of Technology, Masaryk University, Brno, Czech Republic. “We therefore wanted to design, validate, and implement a new custom-designed NGS panel for the integrative analysis of diagnostic, prognostic, and predictive markers.”
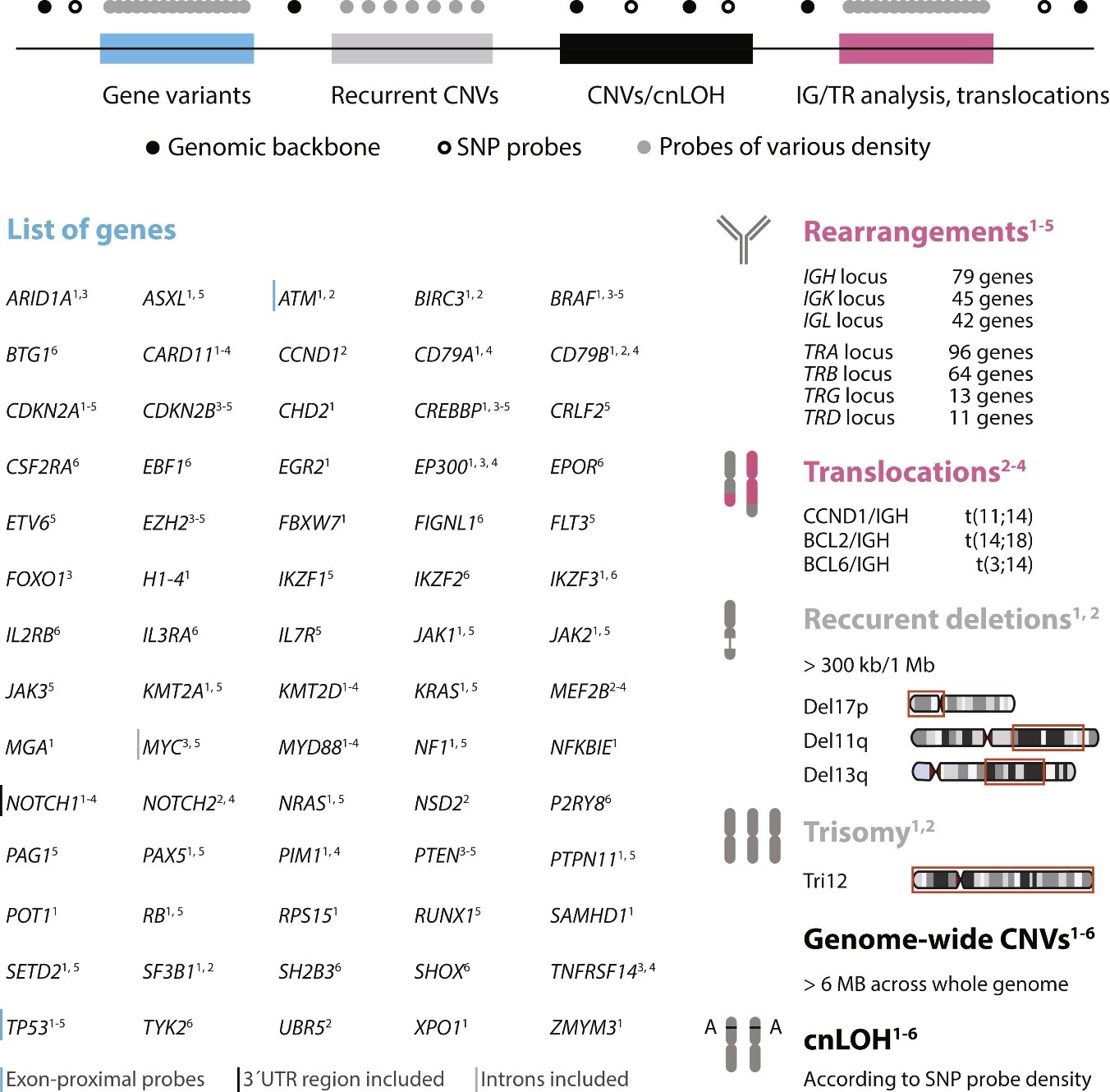
The investigators compiled a list of key genomic biomarkers in chronic lymphocytic leukemia (CLL), acute lymphoblastic leukemia (ALL), diffuse large B-cell lymphoma (DLBCL), follicular lymphoma (FL), and mantle cell lymphoma (MCL) from published and publicly available resources and established a comprehensive NGS panel for their analysis both in routine clinical practice and in biomedical research.
“Such an all-in-one test covering a broad spectrum of crucial biomarkers in lymphoproliferative disorders represents a unique tool for obtaining relevant information about the patient-specific genetic background from just one biological sample,” noted first author Veronika Navrkalova, PhD, from the Faculty of Medicine, Masaryk University and University Hospital Brno; and Central European Institute of Technology, Masaryk University, Brno, Czech Republic.
Caption: A schematic presentation of genomic targets and molecular markers integrated within the LYmphoid NeXt-generation sequencing (LYNX) panel design for the most common lymphoid malignant tumors (1 chronic lymphocytic leukemia, 2 mantle cell lymphoma, 3 follicular lymphoma, 4 diffuse large B-cell lymphoma, 5 acute lymphoblastic leukemia, and 6 Philadelphia chromosome–like acute lymphoblastic leukemia). cnLOH, copy neutral loss of heterozygosity; CNV, copy number variant; SNP, single-nucleotide polymorphism (Credit: The Journal of Molecular Diagnostics).
To ensure the accuracy of the test, researchers validated the reliability of the analytical procedure, which enabled an unbiased identification of various prognostic and predictive markers in a single test. In total, 84 DNA samples from 65 patients (30 with CLL, 13 with ALL, 9 with DLBCL, 6 with MCL, and 7 with FL) were sequenced in two validation rounds. The validation sample cohort was carefully selected to obtain a representative set of different mutation types, copy number variants, common lymphoma translocations, and immunoglobulin/T-cell receptor rearrangements.
A single LYNX test provides accurate detection of mutations in 70 lymphoma-related genes with high sensitivity, reliable identification of large genome-wide and recurrent chromosomal aberrations, the assessment of immunoglobulin and T-cell receptor gene rearrangements, and lymphoma-specific translocation detection.
“This represents a crucial step toward the effective management of hemato-oncological patients,” commented Prof. Dr. Pospisilova. “Because this assay is straightforward and can also be used in research, it warrants further prospective testing in close cooperation among researchers, clinical hemato-oncologists, and hematopathologists to demonstrate its clinical utility and benefit for patients with lymphoid malignant tumors.”
The research team believes that the LYNX panel is suitable for routine testing with research and clinical applicability and may assist in personalized management of patients with lymphoid malignancies. It enables an integrated analysis of clinically relevant genomic aberrations and markers into one test, monitors clonal evolution of the disease, and reveals various genetic architecture in different lymphoproliferative disorders. Furthermore, the results obtained by this test can guide clinical assessment of the patient diagnosis, prognosis, therapy selection and may lead to the revelation of patient-specific markers crucial for monitoring minimal residual disease.
Notes for editors
The article is “LYmphoid NeXt-Generation Sequencing (LYNX) Panel: A Comprehensive Capture-Based Sequencing Tool for the Analysis of Prognostic and Predictive Markers in Lymphoid Malignancies,” by Veronika Navrkalova, Karla Plevova, Jakub Hynst, Karol Pal, Andrea Mareckova, Tomas Reigl, Hana Jelinkova, Zuzana Vrzalova, Kamila Stranska, Sarka Pavlova, Anna Panovska, Andrea Janikova, Michael Doubek, Jana Kotaskova, and Sarka Pospisilova (https://doi.org/10.1016/j.jmoldx.2021.05.007). It appears in The Journal of Molecular Diagnostics, volume 23, issue 8 (August 2021) published by Elsevier.
The article is openly available at https://www.jmdjournal.org/article/S1525-1578(21)00134-3/fulltext.
This work was supported by the projects AZV NV19-03-00091 and Conceptual development of research organization (FNBr 65269705) provided by the Ministry of Health of the Czech Republic, by the research project TACR TJ02000133, GACR GA19-15737S and by the European Regional Development Fund-Project "A-C-G-T "(No. CZ.02.1.01/0.0/0.0/16_026/0008448).
Full text of this study is also available to credentialed journalists upon request; contact Eileen Leahy at +1 732 238 3628 or jmdmedia@elsevier.com. Journalists wishing to interview the authors should contact Prof. Dr. Sarka Pospisilova at sarka.pospisilova@ceitec.muni.cz.
About The Journal of Molecular Diagnostics
The Journal of Molecular Diagnostics, the official publication of the Association for Molecular Pathology, co-owned by the American Society for Investigative Pathology, and published by Elsevier, seeks to publish high quality original papers on scientific advances in the translation and validation of molecular discoveries in medicine into the clinical diagnostic setting, and the description and application of technological advances in the field of molecular diagnostic medicine. The editors welcome review articles that contain: novel discoveries or clinicopathologic correlations, including studies in oncology, infectious diseases, inherited diseases, predisposition to disease, or the description of polymorphisms linked to disease states or normal variations; the application of diagnostic methodologies in clinical trials; or the development of new or improved molecular methods for diagnosis or monitoring of disease or disease predisposition. www.jmdjournal.org
About Elsevier
As a global leader in information and analytics, Elsevier helps researchers and healthcare professionals advance science and improve health outcomes for the benefit of society. We do this by facilitating insights and critical decision-making for customers across the global research and health ecosystems.
In everything we publish, we uphold the highest standards of quality and integrity. We bring that same rigor to our information analytics solutions for researchers, health professionals, institutions and funders.
Elsevier employs 8,100 people worldwide. We have supported the work of our research and health partners for more than 140 years. Growing from our roots in publishing, we offer knowledge and valuable analytics that help our users make breakthroughs and drive societal progress. Digital solutions such as ScienceDirect, Scopus, SciVal, ClinicalKey and Sherpath support strategic research management, R&D performance, clinical decision support, and health education. Researchers and healthcare professionals rely on our 2,500+ digitized journals, including The Lancet and Cell; our 40,000 eBook titles; and our iconic reference works, such as Gray's Anatomy. With the Elsevier Foundation and our external Inclusion & Diversity Advisory Board, we work in partnership with diverse stakeholders to advance inclusion and diversity in science, research and healthcare in developing countries and around the world.
Elsevier is part of RELX, a global provider of information-based analytics and decision tools for professional and business customers. www.elsevier.com.
Media contacts
Eileen Leahy
Elsevier
+1 732 238 3628
Chhavi Chauhan, PhD
Director of Scientific Outreach
The Journal of Molecular Diagnostics
+1 240 283 9724
Source: Elsevier.com


 Share
Share