9. Apr. 2026
CEITEC MUNI contributes to setting an international standard for research on intrinsically disordered proteins
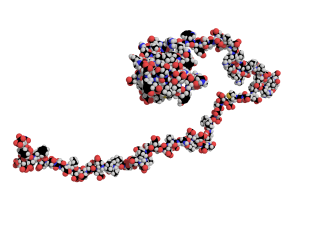
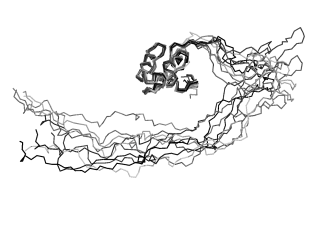
Until about twenty years ago, scientists often dismissed them as “experimental problems” or “artefacts.” They were simply too chaotic and did not fit the prevailing view of proteins as molecules with a stable three-dimensional structure that determines their function. Today, however, research on intrinsically disordered proteins (IDPs) is one of the fastest-growing fields in molecular biology. Surprisingly, around one third of all proteins in the human body belong to this category, playing essential roles in how cells function. Yet they have one defining feature that long hindered their study: they lack a fixed structure and constantly change shape. For this reason, they remained overlooked for many years.
Proteins That Break the Rules of Classical Biology
Most well-known proteins act like precisely engineered molecular machines, with clearly defined shapes linked to specific functions. Intrinsically disordered proteins are different – they behave more like spaghetti than traditional molecular “machines.” They cannot be crystallised, are difficult to visualise using electron microscopy, and appear to challenge the structural principles of biochemistry.
This does not make them any less important. On the contrary, they are involved in the regulation of gene expression, cellular signalling, and processes whose disruption is linked to cancer and neurodegenerative diseases. A well-known example is α-synuclein. This protein does not have a fixed structure and can adopt different shapes depending on the context. One of these forms is associated with Parkinson’s disease, which is commemorated each year on 11 April. To understand the behaviour of proteins such as α-synuclein, entirely new approaches are required.
Brno Expertise in a Global Effort
Researchers Pavel Kadeřávek and Lukáš Žídek from CEITEC Masaryk University (MUNI) are part of a broad international community involving more than thirty institutions worldwide. Together, the researchers have developed a comprehensive framework for studying these elusive proteins. Their recommendations, published in the prestigious journal Nature Methods, summarise current experimental and computational approaches and establish a shared “language” that enables scientists to compare data, combine techniques, and work in a more coordinated way.
CEITEC MUNI plays an important role in this effort thanks to its cutting-edge expertise in nuclear magnetic resonance (NMR) spectroscopy and computational methods – among the few techniques capable of capturing the behaviour of such flexible proteins.
“For a long time, it was believed that a protein without a stable structure could not function. We now know that such proteins are abundant in the human body – and that they carry out some of the most important regulatory roles,” explains Lukáš Žídek from the Josef Dadok National NMR Centre at CEITEC MUNI. “We just need to study them differently.”
Why Future Medicine Depends on These Dynamic Proteins
A better understanding of intrinsically disordered proteins means a better understanding of how certain cancers develop, why proteins form pathological aggregates – as seen in Alzheimer’s or Parkinson’s disease – and how new drugs can be designed to target highly dynamic molecules.
The new international framework will help science move forward in a more unified, faster, and reliable way. And CEITEC MUNI is part of this effort.


 Share
Share